- Elements
Ruthenium
Ruthenium (Ru) is a metal typically found in catalysis, electrochemistry and electrical applications.
XPS analysis of ruthenium is complicated due to the main Ru(3d) core-level overlapping with the C(1s) region, so spectra can be dominated by adventitious carbon signals, or that of an activated carbon support.
As with many metals, Ru exhibits asymmetry to its metallic core-lines, with RuO2 also exhibiting asymmetric Ru(3d) and also O(1s) core levels due to the conducting nature of the material (as shown below).
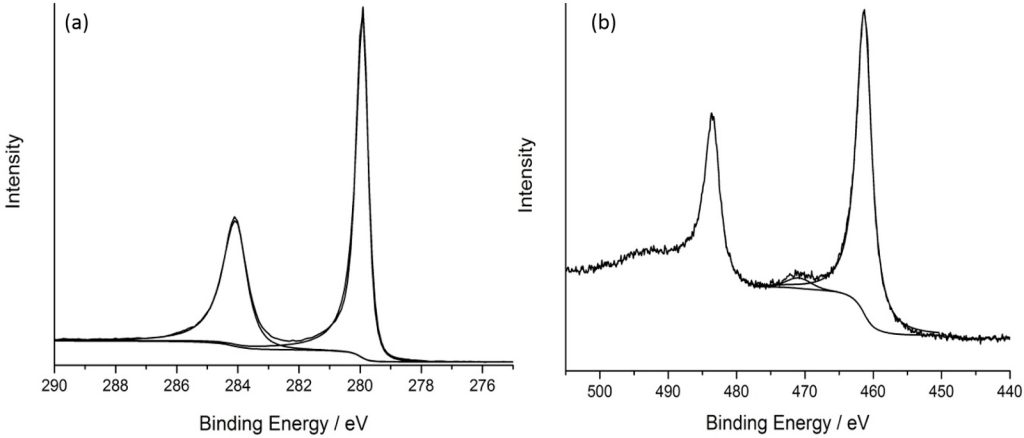
RuO2 is found typically in one of two forms; anhydrous and the more common hydrated form which clearly shows the asymmetric nature of the peaks and screened states giving rise to satellite species often confused with higher oxidation states and can be seen below, together with the adventitious carbon overlap.
