- XPS Theory
The Auger, or Auger-Meitner, Process and Auger Peaks
Recently a movement has begun to rename this process the Auger-Meitner effect, in order to recognise the independent contribution of Lise Meitner in discovering this effect. Though it is traditionally known as simple the Auger effect, we have presented this under both names in order to avoid confusion in readers and attempt to persuade the community to adopt the updated nomenclature.
The Auger process involves the filling of an inner-shell vacancy which results in the emission of an electron from within the same atom. The Auger process takes place in three steps:
- Photoemission of a core electron to leave a core hole (ionization)
- A radiationless transition where an electron from a higher orbital falls to fill the core-hole (relaxation)
- The excess energy of the exited state ion is removed by the ejection of an Auger electron (emission)

Due to this three elcetron process, the shapes of Auger peaks are typiclly more complex than those of most core-levels. An example of an Auger peak is shown below for the MNN Auger peaks (see insert) for metallic Sb, together with the core-level peaks.
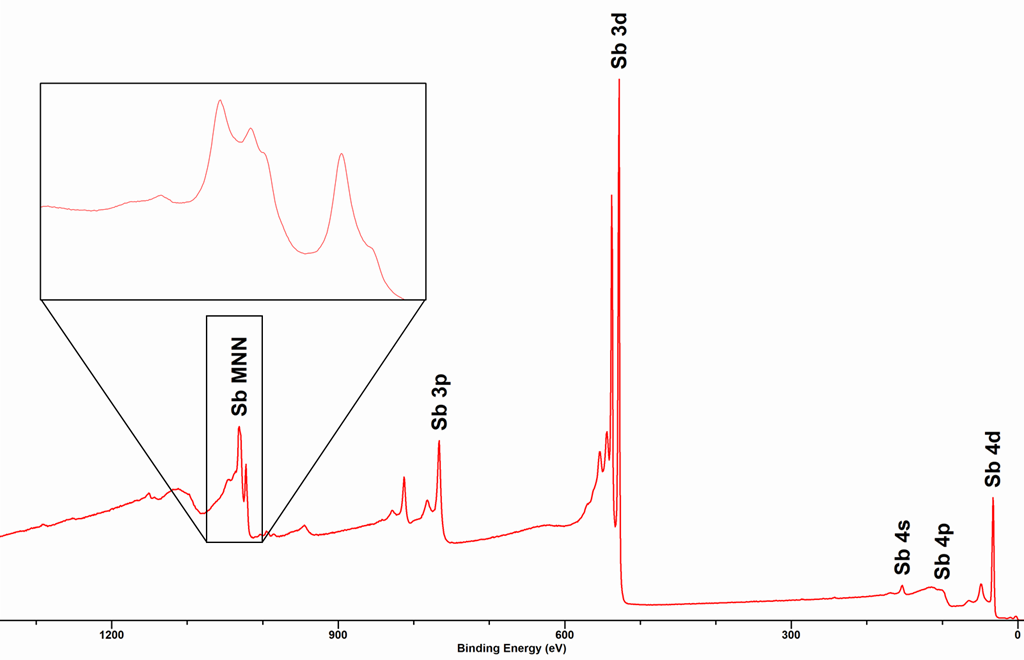
Note that Auger transitions are typically reported a kinetic energy not binding energy and use X-ray notation (K,L,M,N) to state the orbitals involved in the Auger process, these are termed as follows:
| Auger Notation | XPS Notation |
| K | 1s1/2 |
| L1 | 2s1/2 |
| L2 | 2p1/2 |
| L3 | 2p3/2 |
| M1 | 3s1/2 |
| M2 | 3p1/2 |
| M3 | 3p3/2 |
| M4 | 3d3/2 |
| M5 | 3d5/2 |
Notation for Augers involving 4s orbitals and above are detailed in the “Handbook of Applied Solid State Spectroscopy” by Springer